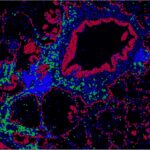
Marceaux C, Tarasova I, Batey D, Yokote K, Gayevskiy V, Jain R, Leiwe M, Choux L, Riley L, Ribera NT, Yang E, Hywood J, Christie M, Antippa P, Speed TP, Rogers KL, Phipson B, Asselin-Labat M-L. TNF-⍺-mediated myeloid-instructed CD14+CD4+ T cells are associated with poor survival in lung adenocarcinoma. Cell Reports Medicine. 2026;7(2):10.1016/j.xcrm.2026.102593
How YY, Reyne N, Croughan MK, Cmielewski P, Batey D, Costello LF, Smith R, Ahlers JN, Cholewa M, Kolodziej M, Duerr J, Mall MA, Kitchen MJ, Asselin-Labat M-L, Paganin DM, Donnelley M, Morgan KS. In Vivo 4D X-Ray Dark-Field Lung Imaging in Mice. IEEE Transactions on Medical Imaging. 2026;45(1):10.1109/tmi.2025.3595666
Smith R, Reyne N, Batey D, Eikelis N, Asselin-Labat M-L, Donnelley M. Mapping lung cancer ventilation dynamics using functional imaging and lung mechanics. Disease Models & Mechanisms. 2025;18(12):10.1242/dmm.052559
Balasubramanian A, Marceaux C, Kanwal S, Tarasova I, Batey D, Senko C, Asadi K, Yokote K, Palmer J, Christie M, Gunjur A, Arulananda S, Parakh S, Adams D, Phipson B, Grimmond S, Cebon J, Asselin-Labat M-L, John T. Effects of CVA21, an Oncolytic Virus, in Combination with Pembrolizumab on Immunogenicity and the Tumor Microenvironment in Advanced NSCLC: A Phase I/II Trial.Clinical Cancer Research. 2025;31(22):10.1158/1078-0432.ccr-25-1449
M. Bader S, Scherer L, Bhandari R, Motyer AJ, Cooney JP, Mackiewicz L, Dayton M, Sheerin D, V. L. Romero D, Schaefer J, Pang J, Chen S, Schoffer K, Wang L, Jin X, Batey D, Yip RKH, Zaman I, Rajasekhar P, Gartner MJ, Wilcox S, Whitehead L, Georgy SR, Maluenda A, Davidson KC, Allison CC, Bowden R, Brinkmann K, Asselin-Labat M-L, Phipson B, Tanzer MC, Herold MJ, Samson AL, Vince JE, Strasser A, Pellegrini M, Doerflinger M. Non-apoptotic caspase-8 is critical for orchestrating exaggerated inflammation during severe SARS-CoV-2 infection. Nature Communications. 2025;16(1):10.1038/s41467-025-65098-z
Balasubramanian A, Asselin-Labat M-L. Immune evolution in pre-invasive lung adenocarcinoma. Cancer Cell. 2025;43(6):10.1016/j.ccell.2025.04.005
Asselin-Labat M-L, Ruhland MK, Ferris ST. Editorial: Antigen presentation in cancer immune responses. Frontiers in Immunology. 2025;16:10.3389/fimmu.2025.1558249
Leiwe M, Asselin-Labat M-L. The dynamics of the metastatic niche: Lung alveolar type 2 cell reprogramming and cancer cell stemness. Developmental Cell. 2024;59(18):10.1016/j.devcel.2024.07.014
VanderDoes J, Marceaux C, Yokote K, Asselin-Labat M-L, Rice G, Hywood JD. Using random forests to uncover the predictive power of distance-varying cell interactions in tumor microenvironments. PLOS Computational Biology. 2024;20(6):10.1371/journal.pcbi.1011361
Bhuva DD, Tan CW, Salim A, Marceaux C, Pickering MA, Chen J, Kharbanda M, Jin X, Liu N, Feher K, Putri G, Tilley WD, Hickey TE, Asselin-Labat M-L, Phipson B, Davis MJ. Library size confounds biology in spatial transcriptomics data. Genome Biology. 2024;25(1):10.1186/s13059-024-03241-7