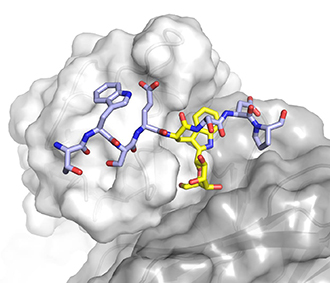
Bloch JS, John A, Mao R, Mukherjee S, Boilevin J, Irobalieva RN, Darbre T, Scott NE, Reymond J-L, Kossiakoff AA, Goddard-Borger ED, Locher KP. Structure, sequon recognition and mechanism of tryptophan C-mannosyltransferase. Nature Chemical Biology. 2023;19(5):10.1038/s41589-022-01219-9
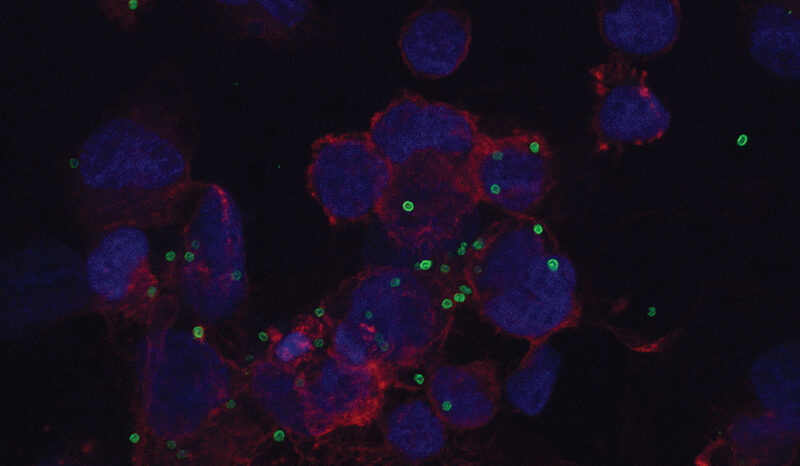
Lopaticki S, McConville R, John A, Geoghegan N, Mohamed SD, Verzier L, Steel RWJ, Evelyn C, O’Neill MT, Soler NM, Scott NE, Rogers KL, Goddard-Borger ED, Boddey JA. Tryptophan C-mannosylation is critical for Plasmodium falciparum transmission. Nature Communications. 2022;13(1):10.1038/s41467-022-32076-8
Sharma M, Lingford JP, Petricevic M, Snow AJD, Zhang Y, Järvå MA, Mui JW-Y, Scott NE, Saunders EC, Mao R, Epa R, da Silva BM, Pires DEV, Ascher DB, McConville MJ, Davies GJ, Williams SJ, Goddard-Borger ED. Oxidative desulfurization pathway for complete catabolism of sulfoquinovose by bacteria. Proceedings of the National Academy of Sciences of the United States of America. 2022;119(4):10.1073/pnas.2116022119
Mao R, Xi S, Shah S, Roy MJ, John A, Lingford JP, Gäde G, Scott NE, Goddard-Borger ED. Synthesis of C‑Mannosylated Glycopeptides Enabled by Ni-Catalyzed Photoreductive Cross-Coupling Reactions. Journal of the American Chemical Society. 2021;143(32):10.1021/jacs.1c05567
John A, Järvå MA, Shah S, Mao R, Chappaz S, Birkinshaw RW, Czabotar PE, Lo AW, Scott NE, Goddard-Borger ED. Yeast- and antibody-based tools for studying tryptophan C-mannosylation. Nature Chemical Biology. 2021;17(4):10.1038/s41589-020-00727-w
Järvå MA, Lingford JP, John A, Soler NM, Scott NE, Goddard-Borger ED. Trefoil factors share a lectin activity that defines their role in mucus. Nature Communications. 2020;11(1):10.1038/s41467-020-16223-7
Järvå MA, Dramicanin M, Lingford JP, Mao R, John A, Jarman KE, Grinter R, Goddard-Borger ED. Structural basis of substrate recognition and catalysis by fucosyltransferase 8. Journal of Biological Chemistry. 2020;295(19):10.1074/jbc.ra120.013291
Lopaticki S, Yang ASP, John A, Scott NE, Lingford JP, O’Neill MT, Erickson SM, McKenzie NC, Jennison C, Whitehead LW, Douglas DN, Kneteman NM, Goddard-Borger ED, Boddey JA. Protein O-fucosylation in Plasmodium falciparum ensures efficient infection of mosquito and vertebrate hosts. Nature Communications. 2017;8(1):10.1038/s41467-017-00571-y
Speciale G, Jin Y, Davies GJ, Williams SJ, Goddard-Borger ED. YihQ is a sulfoquinovosidase that cleaves sulfoquinovosyl diacylglyceride sulfolipids. Nature Chemical Biology. 2016;12(4):10.1038/nchembio.2023
Goddard-Borger ED, Stick RV. An Efficient, Inexpensive, and Shelf-Stable Diazotransfer Reagent: Imidazole-1-sulfonyl Azide Hydrochloride. Organic Letters. 2007;9(19):10.1021/ol701581g