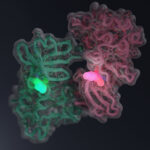
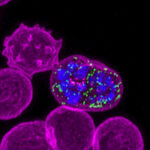
Every molecule in our bodies has a three-dimensional shape, its structure. Structural biology aims to determine the unique shapes of these molecules.
Proteins are relatively large molecules that have an intricate and unique structure. The structure of a protein allows it to fit, or interact, precisely with other molecules, like pieces in a jigsaw puzzle.
Changes in the structure of particular proteins have been linked with many diseases. One cause of these changes to a protein’s structure is alterations (mutations) to the gene that carries the instructions to make that protein.
If genetic mutations occur, the resulting protein structure will be affected. This can alter its ability to interact with other molecules. This affects how the protein functions within the cell, potentially altering the cell’s behaviour. Thus, changes in protein structure can underlie disease.