Diepstraten ST, La Marca JE, Yuan Y, Brown FC, Roberts AW, Wei AH, Kelly GL, Strasser A. Abstract SY35-01: Enhancing BH3 mimetic drug induced killing of malignant cells for cancer therapy. Cancer Research. 2026;86(8_Supplement):10.1158/1538-7445.am2026-sy35-01
Conrad M, Strasser A, Jost PJ, Yuan J, Shao F, Vandenabeele P, Wahida A. Cell death in cancer. Cell. 2026;189(8):10.1016/j.cell.2026.03.024
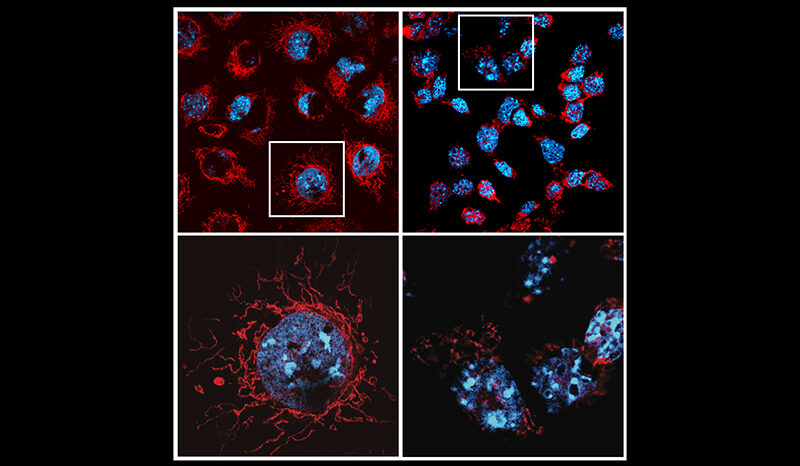
M. Bader S, Scherer L, Bhandari R, Motyer AJ, Cooney JP, Mackiewicz L, Dayton M, Sheerin D, V. L. Romero D, Schaefer J, Pang J, Chen S, Schoffer K, Wang L, Jin X, Batey D, Yip RKH, Zaman I, Rajasekhar P, Gartner MJ, Wilcox S, Whitehead L, Georgy SR, Maluenda A, Davidson KC, Allison CC, Bowden R, Brinkmann K, Asselin-Labat M-L, Phipson B, Tanzer MC, Herold MJ, Samson AL, Vince JE, Strasser A, Pellegrini M, Doerflinger M. Non-apoptotic caspase-8 is critical for orchestrating exaggerated inflammation during severe SARS-CoV-2 infection. Nature Communications. 2025;16(1):10.1038/s41467-025-65098-z
Diepstraten S, La Marca J, Yuan Y, Young S, Chang C, Whelan L, Ross A, Fischer K, Litalien V, Brown F, Roberts A, Wei A, Strasser A, Kelly G. 3151 – PUTTING THE STING BACK INTO THERAPY FOR P53 MUTANT BLOOD CANCERS. Experimental Hematology. 2025;151:10.1016/j.exphem.2025.105092
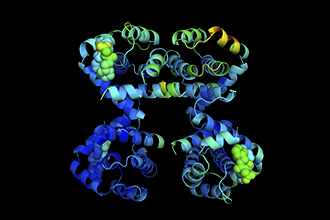
Brinkmann K, McArthur K, Malelang S, Gibson L, Tee A, Elahee Doomun SN, Rowe CL, Arandjelovic P, Marchingo JM, D’Silva D, Bachem A, Monard S, Whelan LG, Dewson G, Putoczki TL, Bouillet P, Fu NY, Brown KK, Kueh AJ, Wimmer VC, Herold MJ, Thomas T, Voss AK, Strasser A. Relative importance of the anti-apoptotic versus apoptosis-unrelated functions of MCL-1 in vivo. Science. 2025;389(6764):10.1126/science.adw1836
Potts MA, Mizutani S, Deng Y, Vaidyanathan S, Ting KE, Giner G, Sridhar S, Shenoy G, Liao Y, Diepstraten ST, Kueh AJ, Pal M, Healey G, Tai L, Wang Z, König C, Kaloni D, Whelan L, Milevskiy MJG, Coughlan HD, Pomilio G, Wei AH, Visvader JE, Papenfuss AT, Wilcox S, Jeyasekharan AD, Shi W, Lelliott EJ, Kelly GL, Brown KK, Strasser A, Herold MJ. Genome-wide in vivo CRISPR screens identify GATOR1 complex as a tumor suppressor in Myc-driven lymphoma. Nature Communications. 2025;16(1):10.1038/s41467-025-62615-y
Gong J-N, Djajawi TM, Moujalled DM, Pomilio G, Khong T, Zhang L-P, Fedele PL, Low MS, Anderson MA, Riffkin CD, White CA, Lan P, Lessene G, Herold MJ, Strasser A, Spencer A, Grigoriadis G, Wei AH, van Delft MF, Roberts AW, Huang DCS. Re-appraising assays on permeabilized blood cancer cells testing venetoclax or other BH3 mimetic agents selectively targeting pro-survival BCL2 proteins. Cell Death & Differentiation. 2025;32(8):10.1038/s41418-025-01487-7
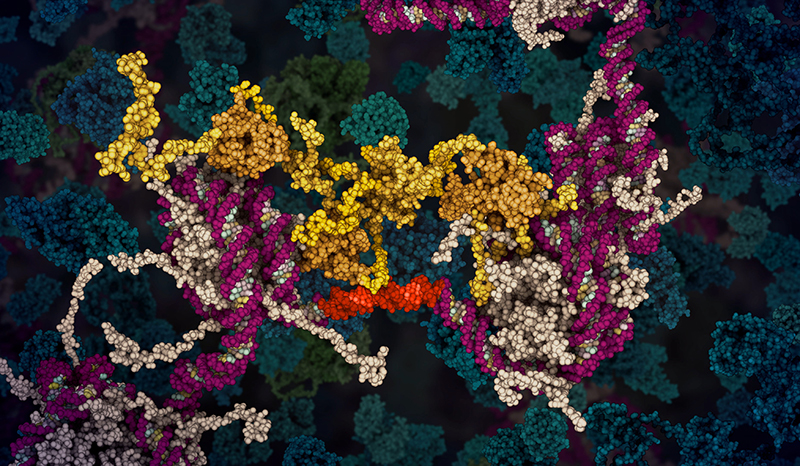
Croce CM, Vaux D, Strasser A, Opferman JT, Czabotar PE, Fesik SW. The BCL-2 protein family: from discovery to drug development. Cell Death & Differentiation. 2025;32(8):10.1038/s41418-025-01481-z
Chin HS, Cheng J, Hsu SH, Lum GG, Zaldiva MT, Guo F, Mallavarapu K, Jackling FC, Smyth GK, Lindeman GJ, Strasser A, Visvader JE, Chen Y, Chen T, Fu NY. Abstract P51: MCL-1 safeguards activated hair follicle stem cells to enable adult hair regeneration. Cancer Research. 2025;85(15_Supplement):10.1158/1538-7445.fcs2024-p51
M. Bader S, Scherer L, Schaefer J, Cooney JP, Mackiewicz L, Dayton M, Georgy SR, Davidson KC, Allison CC, Herold MJ, Strasser A, Pellegrini M, Doerflinger M. IL-1β drives SARS-CoV-2-induced disease independently of the inflammasome and pyroptosis signalling. Cell Death & Differentiation. 2025;32(7):10.1038/s41418-025-01459-x