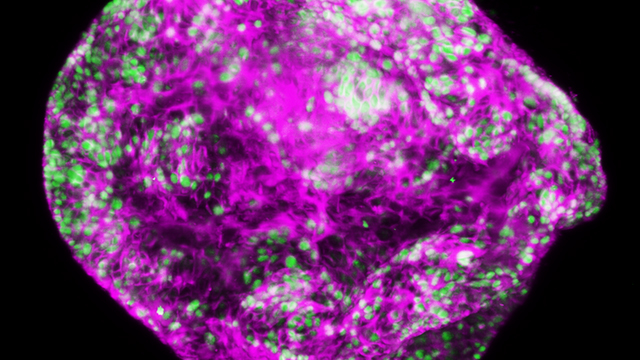
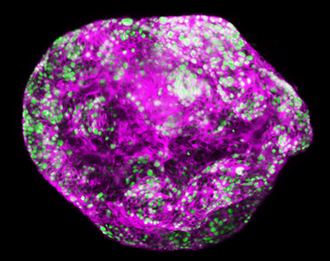
Stomach cancer is uncontrolled growth of cells in the stomach lining. Most stomach cancers are of a type called ‘gastric adenocarcinoma’. This indicates they arise from cells that normally secrete digestive substances into the stomach.
Occasionally cancers in the stomach may arise from immune cells or other tissue types.
More than half of stomach cancers occur in people who have inflammatory conditions in their stomach, termed gastritis. These inflammatory conditions are frequently caused by infection with the bacterium Helicobacter pylori. Our researchers are discovering how inflammation in the stomach lining promotes cancer.
Stomach cancers begin as a tumour within the stomach lining. As this grows it can invade through the stomach wall. Stomach cancers at these early stages are rarely detected, because they cause only mild indigestion-like symptoms.
As stomach cancer develops further, it can spread to nearby lymph nodes. Some cells develop the ability to spread to other organs, called metastasis.
Later stages of stomach cancer can impede the function of the digestive tract and other organs. Symptoms may include:
- Digestive troubles such as vomiting, diarrhoea or constipation.
- Difficulty swallowing.
- Bleeding into the digestive tract.
- Anaemia, from loss of blood.
- Weight loss, weakness and fatigue.
Stomach cancer is the sixth most common cancer diagnosed worldwide. In Australia, more than 2200 cases are diagnosed annually.
Most people with stomach cancer are diagnosed with advanced disease. Current options for treating these people are rarely effective. This contributes to stomach cancer being the second-highest cause of cancer-related deaths globally. More than 1000 Australians die each year from stomach cancer.