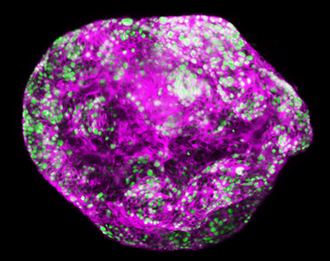
We have specific expertise in Interleukin (IL)-11, a member of the IL-6 family of pleiotropic cytokines. IL-11 signalling is initiated following binding to its membrane bound, cell-type specific receptor, IL-11R. This binary complex engages a transmembrane receptor called GP130, inducing dimerisation and activation of numerous transcription factors, including STAT3. STAT3 has been implicated in the maintenance of a tumour-promoting microenvironment, and persistent STAT3 activation is a feature of many human cancers of both haematopoietic and epithelial origin.
We are part of a multi-institutional collaborative project that was the first to solve the structure of the human IL-11 ligand, receptor and the authentic signalling complex. We build on this knowledge to understand the evolution of cytokine signalling, relationships to disease, and to enable therapeutic development.