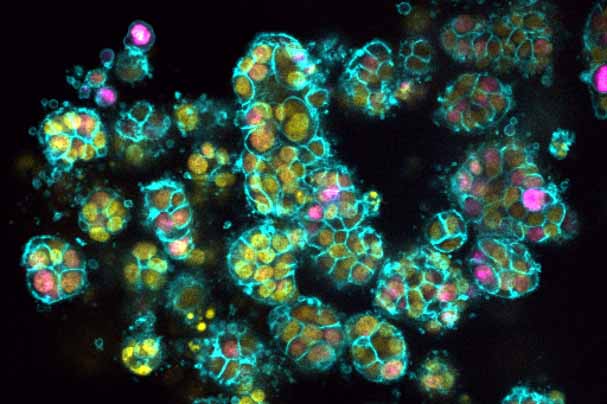
Pires S, Yang W, Frigerio S, Louis C, Scott C, Zhou YL, Cardakli E, Tran N, Hassan-Zahraee M, Ye Z, Hyde C, Hung K, Chen A, Ng C, Grier A, Bank JLC, Lukin D, Scherl E, Targan SR, Diehl GE, Grootjans J, Putoczki TL, Wicks I, Longman RS. Innate lymphoid cells activated by the cytokine TL1A link colitis to emergency granulopoiesis and the recruitment of tumor-promoting neutrophils. Immunity. 2026;59(2):10.1016/j.immuni.2025.12.008
Chiou S, Horne CR, Patel KM, Preaudet A, Rickard JA, Young SN, Jois A, Garnish SE, Hempel A, Hall C, Hildebrand JM, Kueh AJ, Silke J, Putoczki TL, Hawkins ED, Samson AL, Murphy JM. The kinase domain of RIPK3 tunes its scaffolding functions. Cell Death & Differentiation. 2026;:10.1038/s41418-026-01677-x
Brinkmann K, McArthur K, Malelang S, Gibson L, Tee A, Elahee Doomun SN, Rowe CL, Arandjelovic P, Marchingo JM, D’Silva D, Bachem A, Monard S, Whelan LG, Dewson G, Putoczki TL, Bouillet P, Fu NY, Brown KK, Kueh AJ, Wimmer VC, Herold MJ, Thomas T, Voss AK, Strasser A. Relative importance of the anti-apoptotic versus apoptosis-unrelated functions of MCL-1 in vivo. Science. 2025;389(6764):10.1126/science.adw1836
Doggett K, Morgan KJ, Olthof AM, Mieruszynski S, Williams BB, Garnham AL, Milevskiy MJG, Whitehead L, Coates J, Buchert M, O’Donoghue RJJ, Hall TE, Putoczki TL, Ernst M, Sutherland KD, Kanadia RN, Heath JK. Inhibition of the minor spliceosome restricts the growth of a broad spectrum of cancers. EMBO Reports. 2025;26(15):10.1038/s44319-025-00511-8
Zuiani J, Wu D, Roach M, Putoczki T, Saad M, Martelotto L, Ryan F, Drogemuller C, Perkins G, Coates T. 11-3: UNDERSTANDING HEREDITARY PANCREATITIS: PATIENT ORGANOIDS COMPLLIMENT SPATIAL TRANSCRIPTOMIC ANALYSIS. Transplantation. 2025;109(6S1):10.1097/01.tp.0001123200.36575.64
M. Bader S, Calleja DJ, Devine SM, Kuchel NW, Lu BGC, Wu X, Birkinshaw RW, Bhandari R, Loi K, Volpe R, Khakham Y, Au AE, Blackmore TR, Mackiewicz L, Dayton M, Schaefer J, Scherer L, Stock AT, Cooney JP, Schoffer K, Maluenda A, Kleeman EA, Davidson KC, Allison CC, Ebert G, Chen G, Katneni K, Klemm TA, Nachbur U, Georgy SR, Czabotar PE, Hannan AJ, Putoczki TL, Tanzer M, Pellegrini M, Lechtenberg BC, Charman SA, Call MJ, Mitchell JP, Lowes KN, Lessene G, Doerflinger M, Komander D. A novel PLpro inhibitor improves outcomes in a pre-clinical model of long COVID. Nature Communications. 2025;16(1):10.1038/s41467-025-57905-4
Sentosa DD, Metcalfe RD, Sims NA, Putoczki TL, Griffin MDW. The structure of the IL‐11 signalling complex provides insight into receptor variants associated with craniosynostosis. The FEBS Journal. 2025;292(3):10.1111/febs.17307
Pires S, Yang W, Louis C, Tran N, Hassan-Zahraee M, Ye Z, Hung KE, Hyde C, Lukin DJ, Scherl EJ, Targan S, Putoczki T, Wicks I, Longman RS. 220 TL1A DRIVES ILC-MEDIATED GRANULOPOIESIS AND COLITIS ASSOCIATED CANCER. Gastroenterology. 2024;166(5):10.1016/s0016-5085(24)00600-0
Dagley LF, Yousef J, Preaudet A, Loving A, Webb AI, Ernst M, Putoczki TL. Identification of Serum Biomarkers to Monitor Therapeutic Response in Intestinal-Type Gastric Cancer. International Journal of Molecular Sciences. 2024;25(6):10.3390/ijms25063129
Low RRJ, Fung KY, Dagley LF, Yousef J, Emery-Corbin SJ, Putoczki TL. Unbiased Quantitative Proteomics of Organoid Models of Pancreatic Cancer. Methods in Molecular Biology. 2024;2823:10.1007/978-1-0716-3922-1_6