Kousa AI, Jahn L, Zhao K, Flores AE, Acenas D, Lederer E, Argyropoulos KV, Lemarquis AL, Granadier D, Cooper K, D’Andrea M, Sheridan JM, Tsai J, Sikkema L, Lazrak A, Nichols K, Lee N, Ghale R, Malard F, Andrlova H, Velardi E, Youssef S, Burgos da Silva M, Docampo M, Sharma R, Mazutis L, Wimmer VC, Rogers KL, DeWolf S, Gipson B, Gomes ALC, Setty M, Pe’er D, Hale L, Manley NR, Gray DHD, van den Brink MRM, Dudakov JA. Author Correction: Age-related epithelial defects limit thymic function and regeneration. Nature Immunology. 2026;27(5):10.1038/s41590-026-02489-4
Marapana DS, Lopaticki S, Balan B, Reaksudsan K, Cobbold SA, Geoghegan ND, Khurana S, Scally SW, Hickey P, Jayakrishnan N, Vaibhav V, Spall S, Yousef JM, Dagley LF, Goodman CD, Cozijnsen A, McFadden GI, Rogers KL, Jex A, Komander D, McCarthy JS, Tonkin CJ, Dixon MWA, Cowman AF. GID/CTLH E3 ligase complex control cell fate programs for sexual development of Plasmodium falciparum. Nature Communications. 2026;17(1):10.1038/s41467-026-69183-9
Marceaux C, Tarasova I, Batey D, Yokote K, Gayevskiy V, Jain R, Leiwe M, Choux L, Riley L, Ribera NT, Yang E, Hywood J, Christie M, Antippa P, Speed TP, Rogers KL, Phipson B, Asselin-Labat M-L. TNF-⍺-mediated myeloid-instructed CD14+CD4+ T cells are associated with poor survival in lung adenocarcinoma. Cell Reports Medicine. 2026;7(2):10.1016/j.xcrm.2026.102593
Lewis SM, Berthelet J, Whitehead LW, Rajasekhar P, El-Saafin F, Bell C, Naik S, Merino D, Wimmer VC, Rogers KL. LeGO-3D: 3D imaging of lung metastases and vascularisation using light sheet fluorescence microscopy. npj Imaging. 2025;3(1):10.1038/s44303-025-00111-0
Rutter SF, Kang T, Ryan GF, Shi B, Vella CL, Rajasekhar P, Cutter SW, Hodge AL, Ozkocak DC, Ang C-S, Ratcliffe J, Binger KJ, Foneska P, Mathivanan S, Geoghegan ND, Rogers KL, Olson MF, Atkin-Smith GK, Poon IKH. The formation of the ‘footprint of death’ as a mechanism for generating large substrate-bound extracellular vesicles that mark the site of cell death. Nature Communications. 2025;16(1):10.1038/s41467-025-64206-3
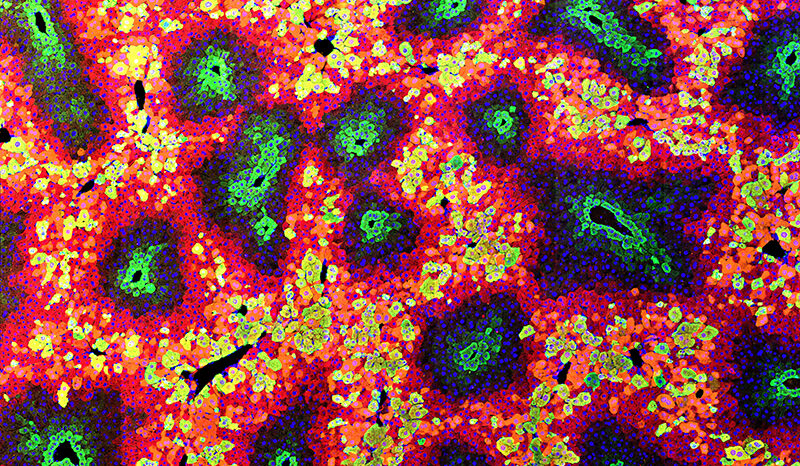
Yip RKH, Er J, Qin L, Nguyen QH, Motyer A, Rimes JS, Light A, Mishi RD, Ling L, Anttila CJA, Tsui E, Amann-Zalcenstein D, Dowling MR, Rogers KL, Bowden R, Chen Y, Harrison SJ, Hawkins ED. Profiling the spatial architecture of multiple myeloma in human bone marrow trephine biopsy specimens with spatial transcriptomics∗†. Blood. 2025;146(15):10.1182/blood.2025028896
Atkin-Smith GK, Santavanond JP, Light A, Rimes JS, Samson AL, Er J, Liu J, Johnson DN, Le Page M, Rajasekhar P, Yip RKH, Geoghegan ND, Rogers KL, Chang C, Bryant VL, Margetts M, Keightley MC, Kilpatrick TJ, Binder MD, Tran S, Lee EF, Fairlie WD, Ozkocak DC, Wei AH, Hawkins ED, Poon IKH. Author Correction: In situ visualization of endothelial cell-derived extracellular vesicle formation in steady state and malignant conditions. Nature Communications. 2025;16(1):10.1038/s41467-025-64770-8
Bryan DJ, Kolesnik K, Szydzik C, Mitchell A, Rogers KL, Collins DJ. Expanding channels enhanced diffractive SAW actuated particle enrichment in vacuum-sealed microfluidic channels. Lab on a Chip. 2025;25(19):10.1039/d4lc00913d
Broomfield BJ, Tan CW, Qin RZ, Abberger H, Duckworth BC, Alvarado C, Dalit L, Lee CL, Mugan RS, Mazrad ZAI, Muramatsu H, Mackiewicz L, Williams BE, Chen J, Takanashi A, Fabb S, Pellegrini M, Rogers KL, Moon WJ, Pouton CW, Davis MJ, Nutt SL, Pardi N, Wimmer VC, Groom JR. Transient inhibition of type I interferon enhances CD8+ T cell stemness and vaccine protection. Journal of Experimental Medicine. 2025;222(5):10.1084/jem.20241148
Bergamasco MI, Ogier JM, Garnham AL, Whitehead L, Rogers K, Smyth GK, Burt RA, Voss AK, Thomas T. Loss of KAT6B causes premature ossification and promotes osteoblast differentiation during development. Developmental Biology. 2025;520:10.1016/j.ydbio.2025.01.012