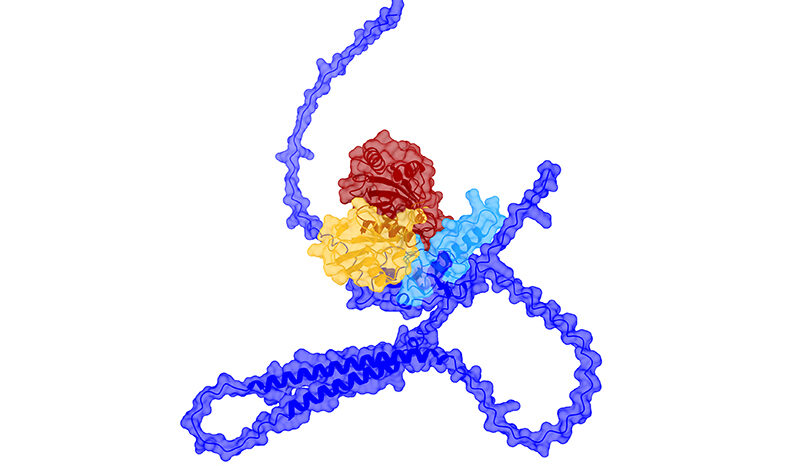
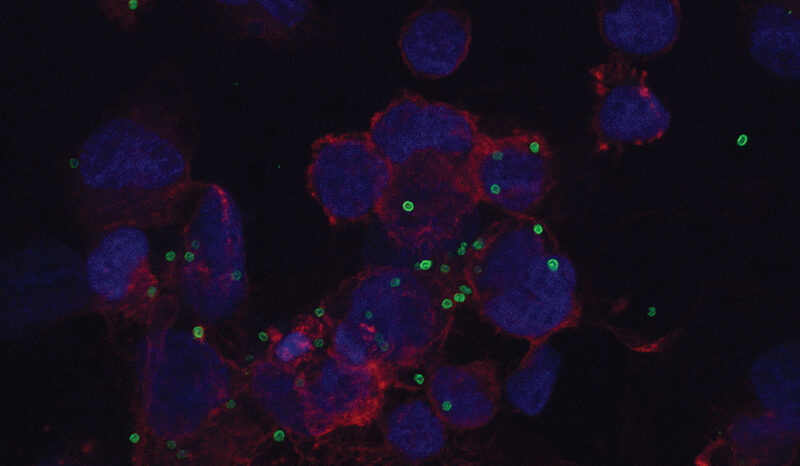
The lab’s earlier work showed that air pollution exposure triggers immune cells in the lung to release an inflammatory signal called interleukin-1 beta (IL-1β), which can wake up dormant cells carrying cancer-causing mutations.
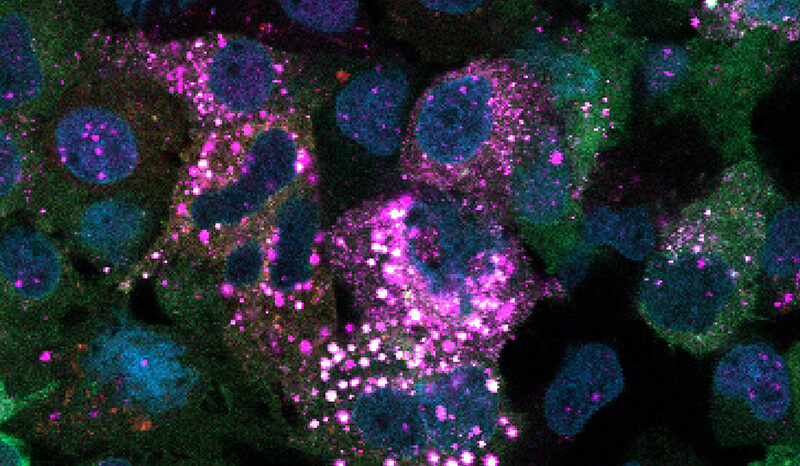
In this study, they showed that pollution exposure simultaneously increases the signature proteins and boosts the population of ‘KAC cells’, an adaptive cell state that occurs in response to injury but can also become cancerous if mutations are present.
The researchers showed that mutant cells from several different lung cell types all enter the same KAC state on their way to cancer, and that air pollution expanded this pool of KAC cells and increased the 14-protein signature.
Dr Weeden said: “What surprised us was that very different lung cell types all seemed to converge on the same intermediate state before becoming cancerous – suggesting KACs represent a common vulnerability in lung adenocarcinoma.
“If we can identify people in whom this process is active and intervene at that stage, we may be able to prevent cancer before it develops.”
The team also found that components of the signature were increased in the presence of IL-1β linked to air pollution.
Blocking IL-1β in mice exposed to pollution reduced the number of KAC cells and slowed early tumour development, suggesting anti-IL-1β drugs could prevent lung cancer in people whose lungs are showing this inflammatory signal.
Tej Pandya, Clinical PhD Student at UCL, said: “Working hand-in-hand with scientists in the lab to understand the biology in mouse models, we’ve shown that the signature reflects an altered inflammatory lung environment before cancer takes hold.
“It’s a proof of concept that, one day, we could use this signature to offer preventive treatment to people at risk of lung cancer.”
In 2017, Novartis’s CANTOS trial tested the IL-1β blocker canakinumab to prevent cardiovascular disease and reported as an exploratory finding that the drug also reduced lung cancer incidence.
However, the benefit was modest at a population level, limiting its use as a prevention strategy in unselected people.
Re-analysing data from 4651 CANTOS participants, the researchers found that people with a high baseline 14-protein signature were the ones who clearly benefited from canakinumab, with their lung cancer risk almost halved.
By selecting only those with a high signature, the number of people needed to treat to prevent one incidence of lung cancer was 55, comparable to established cardiovascular prevention strategies such as statins.
This work was a collaboration between the Francis Crick Institute, UCL, the CRUK Lung Cancer Centre of Excellence, Novartis, RVC, EPIC, ARIC, TALENT and CKB consortia, QMUL, WEHI and the University of Manchester. Funders include the Francis Crick Institute, UCL, CRUK Lung Cancer Centre of Excellence, the Mark Foundation, the Ruth Strauss Foundation, European Research Council, EMBO, Rosetrees Trust, MRC, Wellcome, UKRI and Barts Charity.
–
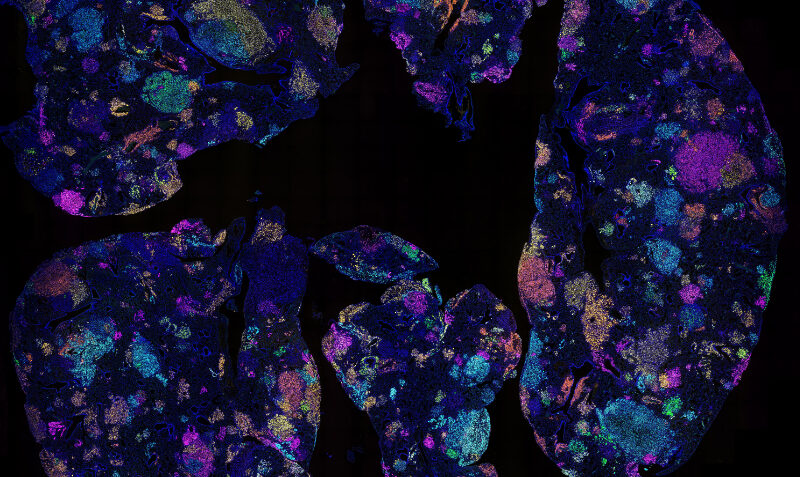
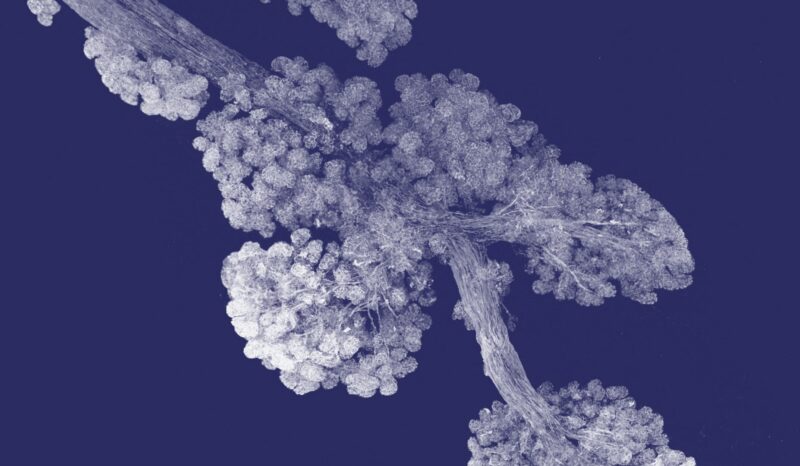
Header image: This image shows tumours growing in a mouse lung, with the colours indicating different gene deletions. The team used mouse models in the study to show the protein signature reflects an altered inflammatory lung environment before cancer takes hold.