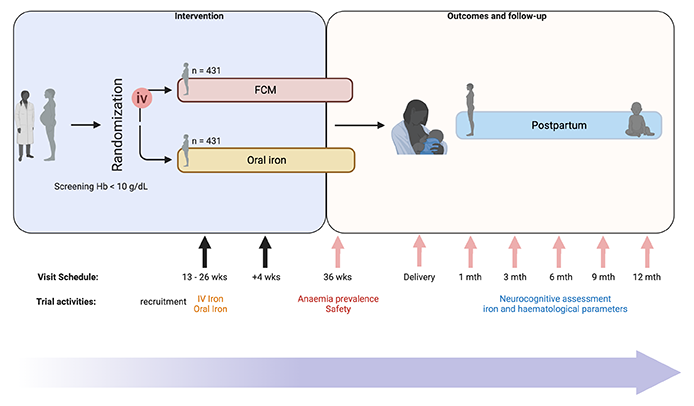
Prof Sant-Rayn Pasricha, Principal Investigator, Population Health and Immunity Division, WEHI
Prof Kamija Phiri, Principal Investigator, Training and Research Unit of Excellence, College of Medicine
Dr Martin Mwangi, Trial Manager, Training and Research Unit of Excellence, College of Medicine
Dr Ricardo Ataide, Trial Manager, Population Health and Immunity Division, WEHI
Ernest Moya, Team Coordinator, Training and Research Unit of Excellence, College of Medicine
Dr Zinani Truwah, Team Coordinator, Training and Research Unit of Excellence, College of Medicine
Dr Glory Mzembe, Team Coordinator, Training and Research Unit of Excellence, College of Medicine
Maclean Vokhiwa, Neuro Assessment Coordinator, Training and Research Unit of Excellence, College of Medicine
Dr Louise Randall, Neuro Study Coordinator, Population Health and Immunity Division, WEHI
Sabine Braat, Senior Trial Statistician, Population Health and Immunity Division, WEHI
Dr Rebecca Harding, Trial Statistician, Population Health and Immunity Division, WEHI
Owen Mtambo, Data Team Leader, Training and Research Unit of Excellence, College of Medicine
Mphatso Mwambinga, Data Officer, Training and Research Unit of Excellence, College of Medicine
Justina Kaunda, Data Officer, Training and Research Unit of Excellence, College of Medicine
Naomi Von Dinklage, Program Coordinator, Population Health and Immunity Division, WEHI