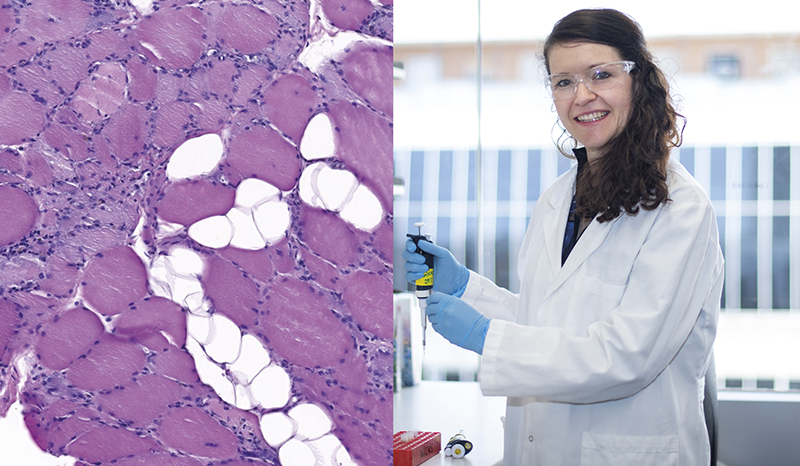
My lab research is focused on molecular analysis of inflammatory and autoimmune diseases. We use murine and human experimental systems and apply ‘omics’ technologies in order to improve understanding of disease pathogenesis, find better biomarkers and develop targeted therapeutics.
Our primary focus has been on rheumatoid arthritis (RA), but we’ve found that experimental systems and techniques used to study RA are relevant to many other inflammatory diseases, and we now have active research programs in vasculitis, myositis and lupus. We also have a strong interest in developing therapeutic antibodies and several of these have gone into clinical trials. In addition to engineered antibodies, we have formed a collaboration with Australian Red Cross Lifeblood to study the naturally occurring human antibody repertoire in hemolytic disease of the newborn.
A major research theme of my lab is to investigate how the cytokines G-CSF and GM-CSF promote inflammatory diseases. This has led to collaborations with the biotechnology sector (CSL, MedImmune and Kiniksa) and the development of antibody-based therapies to the G- and GM-CSF receptors that are currently in clinical trials.