Malaria, says Professor Alan Cowman, is almost the perfect parasite. “Most people who get it do survive,” he says. “The last thing a parasite wants to do is kill its host.”
Even so, malaria kills more than 800,000 people a year, with the death rate higher in small children than adults. In total, the mosquito-borne Plasmodium parasite infects around 250 million people worldwide each year, a huge burden of death, illness and suffering in the developing countries it afflicts.
Taming the malaria parasite
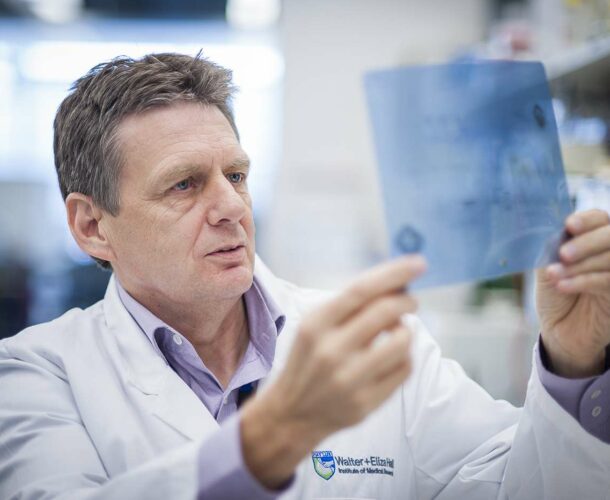
Cowman is a molecular parasitologist and head of the Infection and Immunity division at the Walter and Eliza Hall Institute. He moved from Brisbane to join the institute because “it was the best place in Australia for medical research.”
After more than two decades of working to tame the malaria parasite – “I’ve been working on it since ‘87” – he and his team have produced a potential vaccine with the Seattle Biomedical Research Institute that went through phase 1 clinical trials in Washington, US, with the Walter Reed Army Institute of Research in 2013.
Malaria is a tricky target. Like the common cold virus, it mutates quickly, and developing a vaccine against a moving target is not easy. After it is injected into a person by a mosquito bite, it migrates to the liver, which then releases cells that invade red blood cells, spreading it around the body and causing fever and other symptoms.
“We get red blood cells from the Red Cross and grow parasites on them in a culture dish,” Cowman says. “Then we can do experiments to see how the parasite matures and grows. We can also genetically manipulate them.
“Malaria has been with humans a long time, and there has been a co-evolution. People with sickle-cell anaemia, for example, have a genetic resistance to malaria. All the people in the Mediterranean who have the different thalassaemias (genetic blood disorders that cause anaemia) have some resistance to malaria. Their red cells are altered in a way that doesn’t favour the growth of the parasite.”
Manipulating malaria for vaccine trials
Rather than manipulate human genes, however, Cowman has been experimenting with the genes of the parasite. “The vaccine that reached clinical trial stage is a GAP – a genetically attenuated parasite,” he says. “It contains a live malaria parasite that we have crippled by removing a number of genes out of it.”
Unfortunately, he says, the first trial showed that it was not quite crippled enough. This phase 1 trial was designed to test the vaccine’s safety. Of the six volunteers in Washington injected with the GAP, one actually developed a malaria infection in the blood.
New trials on the horizon
Enter the patience, persistence and analytic skills required of those who use the scientific process. “It didn’t work the way we wanted it to, so we just had to regroup and take one step back,” Professor Cowman says. “So we took one more gene out and there will be new trials this year.
“We are fairly hopeful it will be effective but it’s difficult because it’s a live parasite. There will be all sorts of logistical problems delivering that.”
Programs to eradicate malaria have received a huge boost as a result of the concerns of Melinda Gates, who heads the Bill & Melinda Gates Foundation, a wealthy philanthropic foundation, with her husband, Microsoft billionaire Bill Gates. They recently called for the total elimination of malaria and have said they want to see this happen in their lifetimes.
Focus on eradication
“That has really changed the field,” Professor Cowman says. “It means the vaccines and drugs we are trying to develop have to be geared towards elimination, not just alleviation of disease. Ever since that call, we have really started to change the kind of research we are doing.”
Professor Cowman and his team are collaborating with scientists in other Asia-Pacific countries including Papua New Guinea, Thailand and Indonesia.
This is an international battle, he says. “These sorts of things aren’t just one or two people; they involve large teams across multi-disciplines and in different countries, including the US and Europe. The fight against malaria advances in small steps, which is true of most scientific research.”