A first-in-human clinical trial shows impressive results for treating particularly serious forms of chronic lymphocytic leukaemia (CLL).
CLL is the most common leukaemia in adults, affecting one in 161 Australians by age 85.
Outstanding results
The anti-cancer agent ABT-199 (venetoclax) is used to treat trial participants with advanced leukaemia for whom no conventional treatment options were available.
The treatment achieves outstanding results in a phase one trial including 78 patients with advanced CLL – clearing the cancer in 20 per cent of patients and achieving partial clearance in a further 54 per cent.
Landmark discovery
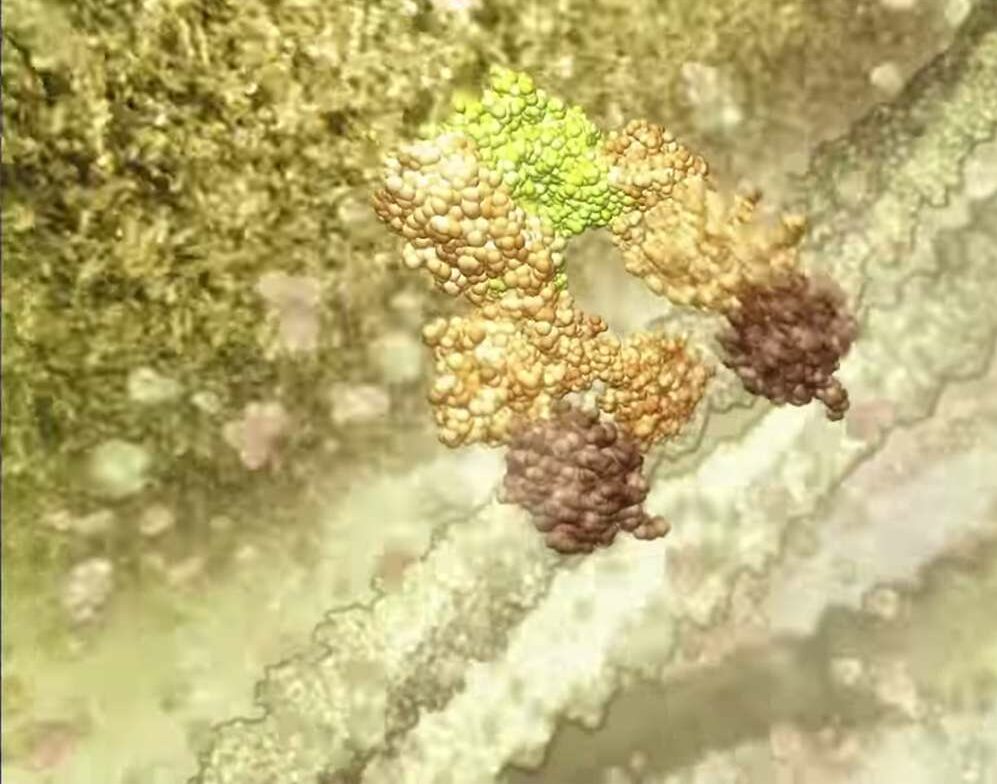
ABT-199 is based on a landmark discovery made in the late 1980s by Walter and Eliza Hall Institute scientists that a protein called Bcl-2 promoted cancer cell survival.
ABT-199/GDC-0199 has been co-developed for clinical use by biotech companies AbbVie and Genentech, a member of the Roche group, and was discovered as part of a joint research collaboration that involved Walter and Eliza Hall Institute scientists. The institute has been closely involved in the preclinical testing and research that led to ABT-199 being used in clinical trials.
Professor Andrew Roberts, a cancer researcher at the institute and haematologist at the Royal Melbourne Hospital, is leading the Melbourne arm of the international trial.
Targeting cancer
Professor Roberts says although CLL cells are slow to proliferate, they accumulated inexorably in the body and, in some patients, result in large tumours that could not be adequately combated with standard treatments.
“High levels of Bcl-2 protect the leukaemia cells from dying when using standard treatments,” Professor Roberts said. “ABT-199 selectively targets the interaction responsible for keeping the leukaemia cells alive and, in many cases, we’ve seen the cancerous cells simply melt away.
“Eighty-four per cent of patients experienced remission after treatment with ABT-199, despite their disease having failed an average of four prior treatment regimens,” he said. “The patients on the trial were typically incurable, with an average life expectancy of up to 18 months. To see complete cancer clearance in nearly one-quarter of these patients, after taking this non-chemotherapy treatment on its own, is incredibly encouraging.”
A Melbourne first
Professor Roberts says it is an important trial and drug for Australia. “We are very happy that our involvement in the discovery has meant that Melbourne patients are some of the first people in the world to benefit from the treatment,” he said.
“Trials are going on around the world now to establish just how important this new drug may be for patients with CLL.”
Potential for future anti-cancer treatments
Studies have suggested the Bcl-2 ‘survival instinct’ may play a role in other cancers, and preclinical testing has suggested drugs that target Bcl-2 could be used to treat other blood cancers, or used in combination with existing treatments to improve treatments for breast, lung and prostate cancers.
The project is an important example of how three Melbourne organisations can collaborate as members of the Victorian Comprehensive Cancer Centre to drive the next generation of improvements in the prevention, detection and treatment of cancer.