As a university student, Dr Ian Majewski worked a summer job in wheat fields, alongside his father, a wheat assessor for Western Australia’s Department of Agriculture. In the 40-degree heat, dust and flies swirling, father and son collected grain samples, which they later cut, threshed and weighed, until, Majewski recalls, “you’d be dreaming wheat”.
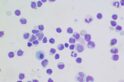
The job helped equip him with the patience and dogged determination required to pursue research into childhood leukaemia, a cancer of the blood, at the Walter and Eliza Hall Institute. Just as he once pored over wheat grains, Majewski now scrutinises thousands of gene sequences for faults— chromosomes may be fused-together, segments of DNA lost, gained or scrambled. He harnesses the power of genetic technologies to pinpoint the faulty genes that drive the disease and resist treatment.
“On the whole, cure rates for childhood leukaemia are good, about 80 per cent of children recover completely,” he explains. “So we’re talking about the 20 per cent or so that are hard to treat.
“The problem is we have such a limited insight into what’s going on with an individual cancer. It might look as if a patient responds well, but then the cancer comes back and it’s clear that something nasty has happened in the genes of the few cancer cells that were leftover.”
The power of genetics
With an honours degree in molecular biology under his belt, Majewski shifted to Melbourne to complete his PhD at the institute, helping Professors Doug Hilton and Warren Alexander identify genes that regulate blood stem cells. With a heightened appreciation of the power of genetics to decipher complex biological systems, a series of fellowships then took him around the world to gain experience with advanced DNA sequencing technology.
In 2009, he began a stint at the Netherlands Cancer Institute, where he studied genes that control cell growth in cancers of the breast and lung. “We had access to a large number of cancer samples with detailed clinical follow-up, which allowed us to find the genes that had the biggest impact on a patient’s outcome,” he explains.
He probes similar questions in his leukaemia research, carried out in conjunction with the Murdoch Childrens Research Institute.
“Right now when a child is diagnosed with leukaemia they’ll be tested, using conventional screening, for three or four common gene mutations,” Majewski explains. “But technological advances mean we can look at every human gene, all 25,000. This gives us a much better understanding of what’s driving the disease.”
Dynamic cancer cells
While we tend to think about evolution and natural selection as something that happens to organisms, in cancer the process operates at the cellular level. Cancer cells are dynamic, Majewski says, always collecting new mutations. These changes can alter the way the cancer responds to treatment. Using a range of techniques, Majewski is comparing the genomes of patients who respond to common chemotherapy drugs to those of patients who fail to respond. Each time he identifies a “good” or “bad” mutation “it’s an excitement tinged with sadness”.
“I keep reminding myself that I have this sample because this patient is unwell. To see this as a rewarding moment you have to focus on the path ahead – the idea that these findings will ultimately provide new ways to treat patients in the future.”
Mutations and other influences
He hopes that by identifying the genetic changes that make cancer cells resistant to chemotherapy, doctors will be able to predict a patient’s response to the drugs and spare them unnecessary and risky treatment. A longer-term aim is the discovery of new treatments that overcome the resistance to chemotherapy.
Still, the complexity of cancer means that genes provide only one set of clues to the fate of individual patients.
“Even when we have all the mutations, there are many other factors that influence cancers, such as changes in gene activity, protein expression, or aspects of the cell’s metabolism. It’s a really complex problem, but we’re learning more every day.”
His project has secured $750,000 funding over five years from the Felton Bequest, including a $500,000 centenary fellowship, a philanthropic grant supporting early-career scientists.